Volume 11, Issue 2 (December 2025)
Elderly Health Journal 2025, 11(2): 135-141 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Mehdipouryan S, Bostani M. Effects of an Eight-Week Aerobic Training Program on Plasma cPLA2 Levels in Sedentary Elderly Men. Elderly Health Journal 2025; 11 (2) :135-141
URL: http://ehj.ssu.ac.ir/article-1-365-en.html
URL: http://ehj.ssu.ac.ir/article-1-365-en.html
Department of Physical Education, Ahv.C., Islamic Azad University, Ahvaz, Iran , mehdi.boustani@iau.ac.ir
Full-Text [PDF 494 kb]
(58 Downloads)
| Abstract (HTML) (234 Views)
Full-Text: (40 Views)
Effects of an Eight-Week Aerobic Training Program on Plasma cPLA2 Levels in Sedentary Elderly Men
Saeideh Mehdipouryan 1, Mehdi Bostani 1*
* Corresponding Author: Department of Physical Education, Ahv.C., Islamic Azad University, Ahvaz, Iran. Tel: +98 9167017668, Email address: mehdi.boustani@iau.ac.ir
Article history
Received 8 Oct 2025
Accepted 30 Nov 2025
A B S T R A C T
Table 1. Aerobic Exercise Protocol
Table 2. Baseline characteristics of the subjects in the two groups
Table 3. The intra-group changes in plasma cPLA2 levels
* Significant difference between pre-test and post-test (p < 0.05)
Table 4. Results of the ANCOVA for between-group comparison
*Significant difference between the two groups
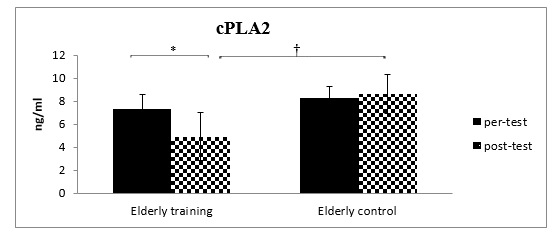
Figure 1. Plasma cPLA2 levels in the aerobic training and control groups before (pre) and after (post) the 8-week intervention. * indicates a significant within-group difference (p < 0.05). † indicates a significant between-group difference (p < 0.05), adjusted for baseline values via ANCOVA
Saeideh Mehdipouryan 1, Mehdi Bostani 1*
- Department of Physical Education, Ahv.C., Islamic Azad University, Ahvaz, Iran
* Corresponding Author: Department of Physical Education, Ahv.C., Islamic Azad University, Ahvaz, Iran. Tel: +98 9167017668, Email address: mehdi.boustani@iau.ac.ir
Article history
Received 8 Oct 2025
Accepted 30 Nov 2025
A B S T R A C T
Introduction: Aging is a gradual, time-dependent process marked by a decline in physiological functions, reduced organ performance, and increased susceptibility to disease. Adopting an active lifestyle can substantially influence both the trajectory and pace of aging. Cytosolic Phospholipase A2 (cPLA2) is an enzyme involved in cell signaling and inflammation through the release of arachidonic acid from membrane phospholipids. The present study aimed to investigate the effects of an eight-week aerobic training program on plasma cPLA2 levels in sedentary elderly men.
Methods: In this quasi-experimental study, twenty elderly men aged 60–74 years were recruited through convenience sampling and randomly assigned to an aerobic training group or a control group. The training group completed an eight-week aerobic exercise program consisting of three sessions per week at 50–75% of heart rate reserve, following the principle of progressive overload. The control group did not engage in any structured physical activity. Blood samples were collected from both groups 48 hours before and after the intervention. Plasma cPLA2 levels were measured using an ELISA kit. Analysis of covariance was used to compare outcomes between groups. Data were analyzed using SPSS version 23 at a significance level of 0.05.
Results: Eight weeks of aerobic training resulted in a significant reduction in plasma cPLA2 levels compared with the control group (p < 0.05).
Conclusion: These findings suggest that regular aerobic exercise may reduce inflammatory markers such as cPLA2 in elderly individuals and may contribute to the management of inflammation-related conditions in older age.
Keywords: Exercise, Phospholipase A2, Aging, Aerobic Training
Copyright © 2025 The Author(s). This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/) which permits copy and redistribute the material just in noncommercial usages, provided the original work is properly cite.
Methods: In this quasi-experimental study, twenty elderly men aged 60–74 years were recruited through convenience sampling and randomly assigned to an aerobic training group or a control group. The training group completed an eight-week aerobic exercise program consisting of three sessions per week at 50–75% of heart rate reserve, following the principle of progressive overload. The control group did not engage in any structured physical activity. Blood samples were collected from both groups 48 hours before and after the intervention. Plasma cPLA2 levels were measured using an ELISA kit. Analysis of covariance was used to compare outcomes between groups. Data were analyzed using SPSS version 23 at a significance level of 0.05.
Results: Eight weeks of aerobic training resulted in a significant reduction in plasma cPLA2 levels compared with the control group (p < 0.05).
Conclusion: These findings suggest that regular aerobic exercise may reduce inflammatory markers such as cPLA2 in elderly individuals and may contribute to the management of inflammation-related conditions in older age.
Keywords: Exercise, Phospholipase A2, Aging, Aerobic Training
Copyright © 2025 The Author(s). This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/) which permits copy and redistribute the material just in noncommercial usages, provided the original work is properly cite.
Introduction
Aging is accompanied by a continuous activation of the immune system, which is observed with high levels of inflammatory markers and activation of immune cells in the circulation and tissues and is associated with an increased risk of many age-related disorders, diseases, and disabilities (1). Cellular senescence is an inevitable process that underlies aging and is associated with age-related diseases such as Alzheimer's disease. Lipids play an important role in cellular aging. Fatty acids, especially eicosanoids, have been linked to various forms of aging and inflammation, and the production of reactive oxygen species associated with it has been proposed as a therapeutic target to reduce aging (2).
The Phospholipase A2 (PLA2) superfamily comprises a large group of extracellular and intracellular enzymes that catalyze the hydrolysis of the sn-2 fatty acyl bond of phospholipids to produce free fatty acids and lysophospholipids. PLA2 constitutes the major metabolic pathway through which fatty acids such as arachidonic acid (AA) are released from their lipid storage sites for the synthesis of eicosanoids, including prostaglandins and leukotrienes (3). Calcium-dependent phospholipase A2 (cPLA2), when activated, catalyzes the conversion of arachidonic acid to eicosanoids such as leukotrienes and prostaglandins. Increased levels of cPLA2 play an important role in several pathways associated with aging, including neuroinflammation and oxidative stress (4). Phospholipases, especially PLA2, are key factors in the membrane hypothesis of aging. In this regard, PLA2 plays an important role in the destabilization of membrane phospholipids, the synthesis of inflammatory mediators, and the production of or response to free radicals. The free fatty acids released by PLA2 are converted to inflammatory mediators such as leukotrienes and prostaglandins, and in this process, free radicals are formed, which in turn can damage further membrane phospholipids. In addition, the lyso-derivatives released by the action of PLA2 have detergent effects that can also damage cell membranes (5). In addition, cPLA2 plays an important role in neuroinflammation by providing precursors for the production of various eicosanoids by cyclooxygenases, lipoxygenases, and prostaglandin synthases (6, 7). Also, cPLA2 activation, which is enhanced by sphingolipid metabolism, enhances the activation of Nuclear factor kappa-light-chain-enhancer of activated B (NF-kB) through interaction with mitochondrial antiviral signaling protein (MAVS) in astrocytes (8). Furthermore, Chuang et al (9) showed that cPLA2 plays a critical role in the signaling of reactive oxygen species (ROS) and nitric oxide (NO) during microglial activation. Furthermore, several studies have focused on identifying the role of cPLA2 in the onset of Alzheimer's disease and have suggested that cPLA2 may be a key player in the pathogenesis of Alzheimer's disease (10-13). Age-related increases in oxidative stress may also contribute to the development of chronic inflammation and inflammatory diseases. It is well established that aging is associated with increased tissue levels and circulation of reactive oxygen species (ROS) as well as decreased antioxidant capacity (14). The role of cPLA2 in mediating inflammatory responses and oxidative stress in microglia, neurons, and astrocytes has been previously demonstrated (15). Several studies have shown that the expression of cPLA2 gene and protein, as well as the levels of its phosphorylated form, are increased in the brains of Alzheimer's patients compared to the brains of healthy individuals (16). The role of exercise as a non-pharmacological and non-complicated environmental intervention in reducing inflammation and increasing antioxidant defenses has been proven (17). Previous studies have shown that regular exercise, especially moderate-intensity exercise, can help reduce inflammation by increasing the body's natural antioxidant levels (18-21). Despite many studies on the effect of exercise on inflammatory markers, the effect of exercise on cPLA2 protein levels during aging has not been studied. Therefore, the aim of the present study was to investigate the effect of eight weeks of aerobic exercise on plasma cPLA2 levels in inactive elderly men.
Methods
Study design and participants
This semi-experimental, applied study examined the effects of an eight-week aerobic training program on plasma cPLA2 levels in sedentary elderly men. The target population consisted of inactive men aged 60–74 years living in Ahvaz, Iran. After screening based on the inclusion criteria, twenty sedentary elderly men were selected through convenience sampling and randomly assigned to one of two groups.
Training group
The training group included ten healthy but inactive elderly men. Blood samples were collected 48 hours before the start of the training protocol. Participants then completed an eight-week aerobic exercise program. A second blood sample was collected 48 hours after the final training session to evaluate changes in cPLA2 levels.
Control group
The control group also consisted of ten healthy, sedentary elderly men. Participants maintained their habitual lifestyle, refrained from any structured exercise, and engaged only in routine daily activities. Blood sampling was performed in parallel with the training group at both pre-test and post-test stages.
Inclusion criteria
Participants met the following criteria:
No engagement in structured sports training for at least six months prior to the study
Age between 60 and 74 years
No use of medications affecting inflammatory markers or cardiovascular function
Non-smokers and non-alcohol users
Absence of cardiovascular disease, hypertension, respiratory disorders, or other chronic health conditions.
Exclusion criteria
Participants were excluded if they experienced injury or illness, withdrew from the study, showed unwillingness to continue the protocol, missed more than three training sessions, or developed symptoms requiring cessation of physical activity (e.g., chest pain), based on (ACSM) guidelines for exercise termination.
Aerobic training protocol
The training program lasted eight weeks and involved three sessions per week. Each 50-minute session comprised a 10-minute warm-up, 30 minutes of aerobic activity, and a 10-minute cool-down. The main exercise was performed at an intensity of 50–75% of heart rate reserve (HRR).
Warm-up and cool-down activities included stretching and light jogging. Participants were instructed to avoid other forms of structured physical activity during the study period.
Training intensity for the first week was set at 50% HRR. Resting heart rate was measured in the supine position, and maximum heart rate was estimated using an age-based formula. Exercise intensity was calculated using the Karvonen formula. Heart rate was continuously monitored using Polar heart rate monitors to ensure adherence to target intensity.
Following the principle of progressive overload, training intensity increased by 5% each week until reaching 75% HRR in weeks seven and eight, in accordance with American College of Sports Medicine guidelines. A summary of the aerobic training progression is shown in table 1.
Blood collection and biochemical analysis
Blood samples were collected from both groups 48 hours before the first training session and 48 hours after the final session by a trained laboratory technician. Samples were obtained after a 12-hour overnight fast while participants were seated and rested. Approximately 5 mL of blood was drawn from the antecubital vein of the left arm into sterile tubes containing EDTA as an anticoagulant.
Samples were centrifuged at 3000 rpm for 10 minutes to separate plasma. Plasma aliquots (1 mL) were transferred to microtubes, transported to the laboratory, and stored at −80°C until analysis. Plasma cPLA2 concentrations were measured using a human ELISA kit (CUSABIO, China) with a sensitivity of 0.11 ng/mL.
Statistical analysis
Data are presented as mean ± standard deviation. The Shapiro–Wilk test was used to assess normality, and Levene’s test was used to evaluate homogeneity of variances. An independent samples t-test was used to compare the basic variables between the two groups. Paired t-tests were performed for within-group comparisons, and analysis of covariance (ANCOVA) was used to compare post-test outcomes between groups while adjusting for baseline values. Statistical analyses were conducted in SPSS version 23, with significance set at p < 0.05.
Ethical considerations
The study protocol was approved by the Ethics Committee of Islamic Azad University, Ahvaz Branch (ID: IR.IAU.AHVAZ.REC.1404.208). Written informed consent was obtained from all participants prior to enrollment.
Results
Descriptive data related to the frequency, mean and standard deviation of age, height, weight and body mass index (BMI) of the subjects in the two groups are presented in Table 2. An independent samples t-test was used to compare the variables between the two groups. As shown in Table 2, the subjects in the two groups were homogeneous in all variables and no significant difference was observed between the two groups.
Intra-group comparison of cPLA2 plasma levels
A paired-samples t-test was used to compare pre- and post-test cPLA2 levels; the results are presented in Table 3. As shown in Table 3, a significant difference was found between pre-test and post-test cPLA2 plasma levels in the elderly training group (p = 0.001), but no significant difference was observed between pre-test and post-test cPLA2 plasma levels in the elderly control group (p = 0.154).
Between-group comparison in plasma cPLA2 levels
Analysis of covariance (ANCOVA) was used to examine the effect of eight weeks of aerobic exercise on plasma cPLA2 levels in inactive elderly men. After confirming the assumptions of this test and adjusting for baseline values, the results of this test are shown in Table 4. As shown in this table, eight weeks of aerobic exercise had a significant effect on plasma cPLA2 levels in inactive elderly men, leading to a significant decrease (p = 0.001). Figure 1 shows the within and Between-group comparison in plasma cPLA2 levels.
The Phospholipase A2 (PLA2) superfamily comprises a large group of extracellular and intracellular enzymes that catalyze the hydrolysis of the sn-2 fatty acyl bond of phospholipids to produce free fatty acids and lysophospholipids. PLA2 constitutes the major metabolic pathway through which fatty acids such as arachidonic acid (AA) are released from their lipid storage sites for the synthesis of eicosanoids, including prostaglandins and leukotrienes (3). Calcium-dependent phospholipase A2 (cPLA2), when activated, catalyzes the conversion of arachidonic acid to eicosanoids such as leukotrienes and prostaglandins. Increased levels of cPLA2 play an important role in several pathways associated with aging, including neuroinflammation and oxidative stress (4). Phospholipases, especially PLA2, are key factors in the membrane hypothesis of aging. In this regard, PLA2 plays an important role in the destabilization of membrane phospholipids, the synthesis of inflammatory mediators, and the production of or response to free radicals. The free fatty acids released by PLA2 are converted to inflammatory mediators such as leukotrienes and prostaglandins, and in this process, free radicals are formed, which in turn can damage further membrane phospholipids. In addition, the lyso-derivatives released by the action of PLA2 have detergent effects that can also damage cell membranes (5). In addition, cPLA2 plays an important role in neuroinflammation by providing precursors for the production of various eicosanoids by cyclooxygenases, lipoxygenases, and prostaglandin synthases (6, 7). Also, cPLA2 activation, which is enhanced by sphingolipid metabolism, enhances the activation of Nuclear factor kappa-light-chain-enhancer of activated B (NF-kB) through interaction with mitochondrial antiviral signaling protein (MAVS) in astrocytes (8). Furthermore, Chuang et al (9) showed that cPLA2 plays a critical role in the signaling of reactive oxygen species (ROS) and nitric oxide (NO) during microglial activation. Furthermore, several studies have focused on identifying the role of cPLA2 in the onset of Alzheimer's disease and have suggested that cPLA2 may be a key player in the pathogenesis of Alzheimer's disease (10-13). Age-related increases in oxidative stress may also contribute to the development of chronic inflammation and inflammatory diseases. It is well established that aging is associated with increased tissue levels and circulation of reactive oxygen species (ROS) as well as decreased antioxidant capacity (14). The role of cPLA2 in mediating inflammatory responses and oxidative stress in microglia, neurons, and astrocytes has been previously demonstrated (15). Several studies have shown that the expression of cPLA2 gene and protein, as well as the levels of its phosphorylated form, are increased in the brains of Alzheimer's patients compared to the brains of healthy individuals (16). The role of exercise as a non-pharmacological and non-complicated environmental intervention in reducing inflammation and increasing antioxidant defenses has been proven (17). Previous studies have shown that regular exercise, especially moderate-intensity exercise, can help reduce inflammation by increasing the body's natural antioxidant levels (18-21). Despite many studies on the effect of exercise on inflammatory markers, the effect of exercise on cPLA2 protein levels during aging has not been studied. Therefore, the aim of the present study was to investigate the effect of eight weeks of aerobic exercise on plasma cPLA2 levels in inactive elderly men.
Methods
Study design and participants
This semi-experimental, applied study examined the effects of an eight-week aerobic training program on plasma cPLA2 levels in sedentary elderly men. The target population consisted of inactive men aged 60–74 years living in Ahvaz, Iran. After screening based on the inclusion criteria, twenty sedentary elderly men were selected through convenience sampling and randomly assigned to one of two groups.
Training group
The training group included ten healthy but inactive elderly men. Blood samples were collected 48 hours before the start of the training protocol. Participants then completed an eight-week aerobic exercise program. A second blood sample was collected 48 hours after the final training session to evaluate changes in cPLA2 levels.
Control group
The control group also consisted of ten healthy, sedentary elderly men. Participants maintained their habitual lifestyle, refrained from any structured exercise, and engaged only in routine daily activities. Blood sampling was performed in parallel with the training group at both pre-test and post-test stages.
Inclusion criteria
Participants met the following criteria:
No engagement in structured sports training for at least six months prior to the study
Age between 60 and 74 years
No use of medications affecting inflammatory markers or cardiovascular function
Non-smokers and non-alcohol users
Absence of cardiovascular disease, hypertension, respiratory disorders, or other chronic health conditions.
Exclusion criteria
Participants were excluded if they experienced injury or illness, withdrew from the study, showed unwillingness to continue the protocol, missed more than three training sessions, or developed symptoms requiring cessation of physical activity (e.g., chest pain), based on (ACSM) guidelines for exercise termination.
Aerobic training protocol
The training program lasted eight weeks and involved three sessions per week. Each 50-minute session comprised a 10-minute warm-up, 30 minutes of aerobic activity, and a 10-minute cool-down. The main exercise was performed at an intensity of 50–75% of heart rate reserve (HRR).
Warm-up and cool-down activities included stretching and light jogging. Participants were instructed to avoid other forms of structured physical activity during the study period.
Training intensity for the first week was set at 50% HRR. Resting heart rate was measured in the supine position, and maximum heart rate was estimated using an age-based formula. Exercise intensity was calculated using the Karvonen formula. Heart rate was continuously monitored using Polar heart rate monitors to ensure adherence to target intensity.
Following the principle of progressive overload, training intensity increased by 5% each week until reaching 75% HRR in weeks seven and eight, in accordance with American College of Sports Medicine guidelines. A summary of the aerobic training progression is shown in table 1.
Blood collection and biochemical analysis
Blood samples were collected from both groups 48 hours before the first training session and 48 hours after the final session by a trained laboratory technician. Samples were obtained after a 12-hour overnight fast while participants were seated and rested. Approximately 5 mL of blood was drawn from the antecubital vein of the left arm into sterile tubes containing EDTA as an anticoagulant.
Samples were centrifuged at 3000 rpm for 10 minutes to separate plasma. Plasma aliquots (1 mL) were transferred to microtubes, transported to the laboratory, and stored at −80°C until analysis. Plasma cPLA2 concentrations were measured using a human ELISA kit (CUSABIO, China) with a sensitivity of 0.11 ng/mL.
Statistical analysis
Data are presented as mean ± standard deviation. The Shapiro–Wilk test was used to assess normality, and Levene’s test was used to evaluate homogeneity of variances. An independent samples t-test was used to compare the basic variables between the two groups. Paired t-tests were performed for within-group comparisons, and analysis of covariance (ANCOVA) was used to compare post-test outcomes between groups while adjusting for baseline values. Statistical analyses were conducted in SPSS version 23, with significance set at p < 0.05.
Ethical considerations
The study protocol was approved by the Ethics Committee of Islamic Azad University, Ahvaz Branch (ID: IR.IAU.AHVAZ.REC.1404.208). Written informed consent was obtained from all participants prior to enrollment.
Results
Descriptive data related to the frequency, mean and standard deviation of age, height, weight and body mass index (BMI) of the subjects in the two groups are presented in Table 2. An independent samples t-test was used to compare the variables between the two groups. As shown in Table 2, the subjects in the two groups were homogeneous in all variables and no significant difference was observed between the two groups.
Intra-group comparison of cPLA2 plasma levels
A paired-samples t-test was used to compare pre- and post-test cPLA2 levels; the results are presented in Table 3. As shown in Table 3, a significant difference was found between pre-test and post-test cPLA2 plasma levels in the elderly training group (p = 0.001), but no significant difference was observed between pre-test and post-test cPLA2 plasma levels in the elderly control group (p = 0.154).
Between-group comparison in plasma cPLA2 levels
Analysis of covariance (ANCOVA) was used to examine the effect of eight weeks of aerobic exercise on plasma cPLA2 levels in inactive elderly men. After confirming the assumptions of this test and adjusting for baseline values, the results of this test are shown in Table 4. As shown in this table, eight weeks of aerobic exercise had a significant effect on plasma cPLA2 levels in inactive elderly men, leading to a significant decrease (p = 0.001). Figure 1 shows the within and Between-group comparison in plasma cPLA2 levels.
Table 1. Aerobic Exercise Protocol
| Week | Frequency (sessions/week) | Duration (min/session) | Intensity (% HRR) |
| 1 | 3 | 50 | 50 |
| 2 | 3 | 50 | 55 |
| 3 | 3 | 50 | 60 |
| 4 | 3 | 50 | 65 |
| 5 | 3 | 50 | 70 |
| 6 | 3 | 50 | 70 |
| 7 | 3 | 50 | 75 |
| 8 | 3 | 50 | 75 |
Table 2. Baseline characteristics of the subjects in the two groups
| Variable | Group | N | Mean ± S.D | Mean difference | df | t | p |
| Age (year) | Training | 10 | 3.47±66.40 | 1.60 |
18 | 1.178 | 0.254 |
| Control | 10 | 2.52 ±64.80 | |||||
| Height (cm) | Training | 10 | 3.02±175.50 | 2.30 | 18 | 1.758 | 0.096 |
| Control | 10 | 2.82 ± 177.80 | |||||
| Weight (Kg) | Training | 10 | 3.83± 84.70 | 0.90 | 18 | 0.520 | 0.609 |
| Control | 10 | 3.91±83.80 | |||||
| BMI (kg/m2) | Training | 10 | 1.16±27.50 | 0.974 | 18 | 1.585 | 0.130 |
| Control | 10 | 1.55±26.52 |
Table 3. The intra-group changes in plasma cPLA2 levels
| p | t | df | difference (Pre - Post) | Mean ± S.D | Stage | Group |
| 0.001* | 8.08 | 9 | 2.07 | 1.61±8.26 | Pre-test | Training |
| 1.13±6.18 | post-test | |||||
| 0.154 | 1.55 | 9 | 0.059 | 1.50 ±8.60 | Pre-test | Control |
| 1.57±8.65 | post-test |
* Significant difference between pre-test and post-test (p < 0.05)
Table 4. Results of the ANCOVA for between-group comparison
| p | F | Mean Square | df | Type III Sum of Squares | |
| 0.001 | 110.629 | 29.946 | 2 | 59.893 | Corrected Model |
| 0.442 | 0.619 | 0.168 | 1 | 0.168 | Intercept |
| 0.336 | 5.131 | 1.972 | 17 | 33.532 | Pre-test |
| 0.001* | 87.908 | 23.796 | 1 | 23.796 | Group |
| 0.271 | 17 | 4.602 | Error | ||
| 20 | 1166.365 | Total | |||
| 19 | 64.495 | Corrected total |
*Significant difference between the two groups

Figure 1. Plasma cPLA2 levels in the aerobic training and control groups before (pre) and after (post) the 8-week intervention. * indicates a significant within-group difference (p < 0.05). † indicates a significant between-group difference (p < 0.05), adjusted for baseline values via ANCOVA
Discussion
Aging is associated with increased inflammation and related diseases (1). Regular exercise is one of the most important strategies for controlling inflammation, especially in older people (23). Therefore, the aim of the present study was to investigate the effect of eight weeks of aerobic exercise on cPLA2 plasma levels.
The results of the present study showed that eight weeks of aerobic exercise significantly reduced plasma cPLA2 levels in inactive elderly men. Very few studies have been conducted on the effect of exercise on cPLA2. Buyukyazi et al. (24) investigated the effect of different walking intensities on PLA2 in postmenopausal women and concluded that 12 weeks of both moderate and high intensity walking resulted in significant reductions in PLA2.
Given that PLA2 enzyme is expressed in atherosclerotic plaques, macrophages, and neutrophils in the endothelium and released into the blood, aerobic exercise can prevent the transformation of monocytes into macrophages and the formation of plaque in the intima by increasing the antioxidant capacity of the intima and reducing the inflammatory response in the long term, and consequently reduce the production of this enzyme at the source (25). On the other hand, since cPLA2 leads to inflammation and down regulation of eNOS in the vascular wall by oxidative modification of LDL and production of lysophosphatidylcholine and non-esterified oxidized fatty acids, it is plausible that the reduction in cPLA2 represents another mechanism through which exercise improves vascular function, given that cPLA2 inhibition has been shown to reduce the number of macrophages and the thickness of the fibrous cap of atherosclerotic plaques (26).
The free radical theory of aging, which is associated with the accumulation of oxidative stress damage, can be a description of the aging process and the signaling network activated by ROS. According to it, the ability of the organism to cope with cellular damage caused by ROS decreases, causing oxidative stress. As a result, aging is associated with oxidative damage to proteins and DNA (27).
Physical activity may affect oxidative stress processes through several mechanisms, including oxygen leakage from the electron transport chain, prostanoid metabolism, xanthine oxidase and macrophage activity, and increased catecholamine activity (28). Exercise exerts antioxidant effects by suppressing inflammatory pathways and inhibiting prominent sources of RONS. Exercise also activates redox-sensitive transcription factors (NF-κB, AP-1, and PGC-1β), leading to enhanced antioxidant defense mechanisms. It is clear that the effects of exercise vary depending on the type, intensity, frequency, and duration of exercise, as well as the age, sex, fitness, and health status of individuals (29).
In normal conditions, the body secretes anti-inflammatory factors against inflammation, but when due to various factors such as metabolic diseases including diabetes, obesity and aging, inflammation becomes out of balance and moves towards pathogenesis. In aging, inflammation is associated with the occurrence of many neurological diseases such as Alzheimer's and dementia, with metabolic diseases such as diabetes, hypertension and metabolic syndrome, as well as with some types of cancer (30). Exercise has both an anti-inflammatory effect through various pathways and reduces the secretion of inflammatory factors by reducing body fat mass and enhancing immune system function (31).
The exact mechanisms by which physical activity reduces inflammation are not fully understood. One of the possible mechanisms for the effects of exercise training on improving the inflammatory state is the intracellular signaling pathways mediated by nitric oxide (NO). Nitric oxide is closely related to the inflammatory state and acts as an important mediator of inflammation. The enzyme nitric oxide synthase (eNOS) regulates the expression of proinflammatory molecules such as NF-κB and cyclooxygenase 2 (COX2) as well as proinflammatory cytokines. Nitric oxide derived from eNOS plays an important role in some of the fundamental events of inflammation such as cell adhesion, modulation of vascular permeability, and angiogenesis. Notably, after moderate-intensity exercise, nitric oxide levels increase, leading to improved muscle function and inhibition of inflammation (32).
This study, conducted on an elderly population, faced inherent limitations regarding sample size, restricting us to 10 participants per group. Other limitations include the lack of measurement of additional inflammatory markers (e.g., TNF-α, IL-6) and the inability to control for potentially confounding environmental factors such as diet, sleep, and stress, which can influence inflammation.
Conclusion
This study demonstrated that an eight-week aerobic exercise program can reduce levels of the inflammatory marker cPLA2, potentially helping to prevent the onset of inflammation-related disorders in aging. Therefore, aerobic exercise can be considered a non-pharmacological strategy for mitigating aging-associated oxidative stress and enhancing the quality of life in the elderly.
Acknowledgements
This article is derived from the master's thesis of the Islamic Azad University of Ahvaz Branch, which hereby acknowledges and thanks the research deputy of the Islamic Azad University of Ahvaz Branch and the research participants.
Conflict of interest
The authors of the article declare that there is no conflict of interest in the present study.
Funding
This research received no external funding.
Authors' Contributions
Conceptualization: S.M., M.B.; Methodology: S.M., M.B.; Formal analysis: S.M., M.B.; Investigation: S.M., M.B.; Writing - original draft: S.M., M.B.; Writing–review & editing: S.M., M.B.; Supervision: M.B. All authors approved the final manuscript.
References
1. Singh A, Schurman SH, Bektas A, Kaileh M, Roy R, Wilson DM, et al. Aging and inflammation. Cold Spring Harbor Perspectives in Medicine. 2024; 14(6): 1-26.
2. Sun GY, Geng X, Teng T, Yang B, Appenteng MK, Greenlief CM, et al. Dynamic role of phospholipases A2 in health and diseases in the central nervous system. Cells. 2021; 10(11): 1-23.
3. Khan SA, Ilies MA. The phospholipase A2 superfamily: structure, isozymes, catalysis, physiologic and pathologic roles. International Journal of Molecular Sciences. 2023; 24(2): 1-35.
4. Hugo C, Asante I, Sadybekov A, Katritch V, Yassine HN. Development of calcium-dependent phospholipase a2 inhibitors to target cellular senescence and oxidative stress in neurodegenerative diseases. Antioxidants & Redox Signaling. 2024; 41(16-18): 1100-16.
5. Mukherjee AK. Correlation between the phospholipids domains of the target cell membrane and the extent of Naja kaouthia PLA2-induced membrane damage: evidence of distinct catalytic and cytotoxic sites in PLA2 molecules. Biochimica et Biophysica Acta (BBA)-General Subjects. 2007; 1770(2): 187–95.
6. Czapski GA, Czubowicz K, Strosznajder JB, Strosznajder RP. The lipoxygenases: their regulation and implication in Alzheimer’s disease. Neurochemical Research. 2016; 41(1): 243–57.
7. Strokin M, Sergeeva M, Reiser G. Docosahexaenoic acid and arachidonic acid release in rat brain astrocytes is mediated by two separate isoforms of phospholipase A2 and is differently regulated by cyclic AMP and Ca2+. British Journal of Pharmacology. 2003; 139(5): 1014-22.
8. Chao CC, Gutiérrez-Vázquez C, Rothhammer V, Mayo L, Wheeler MA, Tjon EC, et al. Metabolic control of astrocyte pathogenic activity via cPLA2-MAVS. Cell. 2019; 179(7): 1483–98.
9. Chuang DY, Simonyi A, Kotzbauer PT, Gu Z, Sun GY. Cytosolic phospholipase A2 plays a crucial role in ROS/NO signaling during microglial activation through the lipoxygenase pathway. Journal of Neuroinflammation. 2015; 12(1): 1-20.
10. Lee JCM, Simonyi A, Sun AY, Sun GY. Phospholipases A2 and neural membrane dynamics: implications for Alzheimer’s disease. Journal of Neurochemistry. 2011; 116(5): 813–9.
11. Gentile MT, Reccia M, Sorrentino P, Vitale E, Sorrentino G, Puca AA, et al. Role of cytosolic calcium-dependent phospholipase A2 in Alzheimer's disease pathogenesis. Molecular Neurobiology. 2012; 45(3): 596–604.
12. Gynther M, Estrada ML, Loppi S, Korhonen P, Kanninen KM, Malm T, et al. Increased expression and activity of brain cortical cPLA2 due to chronic lipopolysaccharide administration in mouse model of familial alzheimer’s disease. Pharmaceutics. 2022; 14(11): 1-12.
13. Ma QL, Ebright B, Li B, Li J, Galvan J, Sanchez A, et al. Evidence for cPLA2 activation in Alzheimer’s disease synaptic pathology. BioRxiv. 2025; 1: 1-43.
14. Kregel KC, Zhang HJ. An integrated view of oxidative stress in aging: basic mechanisms, functional effects, and pathological considerations. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology. 2007; 292(1): 18– 36.
15. Sun GY, Chuang DY, Zong Y, Jiang J, Lee JC, Gu Z, et al. Role of cytosolic phospholipase A2 in oxidative and inflammatory signaling pathways in different cell types in the central nervous system. Molecular Neurobiology. 2014; 50(1): 6–14.
16. Sanchez-Mejia RO, Newman JW, Toh S, Yu GQ, Zhou Y, Halabisky B, et al. Phospholipase A2 reduction ameliorates cognitive deficits in a mouse model of Alzheimer's disease. Nature Neuroscience. 2008; 11(11): 1311–8.
17. Bostani M, Noaein SA. The effect of continuous aerobic training on bax/bcl2 ratio in pancreatic tissue diabetic rats. Iranian Journal of Diabetes and Obesity. 2021.
18. Cerqueira É, Marinho DA, Neiva HP, Lourenço O. Inflammatory effects of high and moderate intensity exercise—a systematic review. Frontiers in Physiology. 2020; 10: 1-14.
19. de Sousa CV, Sales MM, Rosa TS, Lewis JE, de Andrade RV, Simoes HG. The antioxidant effect of exercise: a systematic review and meta-analysis. Sports Medicine. 2017; 47(2): 277–93.
20. de Souza DC, Matos VA, Dos Santos VO, Medeiros IF, Marinho CS, Nascimento PR, et al. Effects of high-intensity interval and moderate-intensity continuous exercise on inflammatory, leptin, IgA, and lipid peroxidation responses in obese males. Frontiers in Physiology. 2018; 9: 1-9.
21. Pillon Barcelos R, Freire Royes LF, Gonzalez-Gallego J, Bresciani G. Oxidative stress and inflammation: liver responses and adaptations to acute and regular exercise. Free Radical Research. 2017; 51(2): 222–36.
22. Bushman B, Medicine ACoS. ACSM's Complete Guide to Fitness & Health, 2E: Human Kinetics .2017.
23. Gómez-Rubio P, Trapero I. The beneficial effect of physical exercise on inflammatory makers in older individuals. Endocrine, Metabolic & Immune Disorders-Drug Targets . 2021; 21(6): 1008-16.
24. Buyukyazi G, Ulman C, Taneli F, Esen H, Gozlukaya F, Ozcan I, et al. The effects of different intensity walking programs on serum blood lipids, high-sensitive C-reactive protein, and lipoprotein-associated phospholipase A2 in premenopausal women. Science & Sports. 2010; 25(5): 245–52.
25. Thijssen DH, Cable NT, Green DJ. Impact of exercise training on arterial wall thickness in humans. Clinical Science. 2012; 122(7): 311–22.
26. Lavi S, McConnell JP, Rihal CS, Prasad A, Mathew V, Lerman LO, et al. Local production of lipoprotein-associated phospholipase A2 and lysophosphatidylcholine in the coronary circulation: association with early coronary atherosclerosis and endothelial dysfunction in humans. Circulation. 2007; 115(21): 2715–21.
27. Vina J, Borras C, Abdelaziz KM, Garcia-Valles R, Gomez-Cabrera MC. The free radical theory of aging revisited: the cell signaling disruption theory of aging. Antioxidants & Redox Signaling. 2013; 19(8): 779–87.
28. Kruk J, Aboul-Enein HY, Kładna A, Bowser JE. Oxidative stress in biological systems and its relation with pathophysiological functions: the effect of physical activity on cellular redox homeostasis. Free Radical Research. 2019; 53(5): 497–521.
29. Webb R, Hughes MG, Thomas AW, Morris K. The ability of exercise-associated oxidative stress to trigger redox-sensitive signalling responses. Antioxidants. 2017; 6(3): 1-21.
30. Deleidi M, Jäggle M, Rubino G. Immune aging, dysmetabolism, and inflammation in neurological diseases. Frontiers in Neuroscience. 2015;9: 1-14.
31. Wang S, Zhou H, Zhao C, He H. Effect of exercise training on body composition and inflammatory cytokine levels in overweight and obese individuals: a systematic review and network meta-analysis. Frontiers in Immunology. 2022; 13: 1-13.
32. Scheele C, Nielsen S, Pedersen BK. ROS and myokines promote muscle adaptation to exercise. Trends in Endocrinology & Metabolism. 2009; 20(3): 95–9.
Aging is associated with increased inflammation and related diseases (1). Regular exercise is one of the most important strategies for controlling inflammation, especially in older people (23). Therefore, the aim of the present study was to investigate the effect of eight weeks of aerobic exercise on cPLA2 plasma levels.
The results of the present study showed that eight weeks of aerobic exercise significantly reduced plasma cPLA2 levels in inactive elderly men. Very few studies have been conducted on the effect of exercise on cPLA2. Buyukyazi et al. (24) investigated the effect of different walking intensities on PLA2 in postmenopausal women and concluded that 12 weeks of both moderate and high intensity walking resulted in significant reductions in PLA2.
Given that PLA2 enzyme is expressed in atherosclerotic plaques, macrophages, and neutrophils in the endothelium and released into the blood, aerobic exercise can prevent the transformation of monocytes into macrophages and the formation of plaque in the intima by increasing the antioxidant capacity of the intima and reducing the inflammatory response in the long term, and consequently reduce the production of this enzyme at the source (25). On the other hand, since cPLA2 leads to inflammation and down regulation of eNOS in the vascular wall by oxidative modification of LDL and production of lysophosphatidylcholine and non-esterified oxidized fatty acids, it is plausible that the reduction in cPLA2 represents another mechanism through which exercise improves vascular function, given that cPLA2 inhibition has been shown to reduce the number of macrophages and the thickness of the fibrous cap of atherosclerotic plaques (26).
The free radical theory of aging, which is associated with the accumulation of oxidative stress damage, can be a description of the aging process and the signaling network activated by ROS. According to it, the ability of the organism to cope with cellular damage caused by ROS decreases, causing oxidative stress. As a result, aging is associated with oxidative damage to proteins and DNA (27).
Physical activity may affect oxidative stress processes through several mechanisms, including oxygen leakage from the electron transport chain, prostanoid metabolism, xanthine oxidase and macrophage activity, and increased catecholamine activity (28). Exercise exerts antioxidant effects by suppressing inflammatory pathways and inhibiting prominent sources of RONS. Exercise also activates redox-sensitive transcription factors (NF-κB, AP-1, and PGC-1β), leading to enhanced antioxidant defense mechanisms. It is clear that the effects of exercise vary depending on the type, intensity, frequency, and duration of exercise, as well as the age, sex, fitness, and health status of individuals (29).
In normal conditions, the body secretes anti-inflammatory factors against inflammation, but when due to various factors such as metabolic diseases including diabetes, obesity and aging, inflammation becomes out of balance and moves towards pathogenesis. In aging, inflammation is associated with the occurrence of many neurological diseases such as Alzheimer's and dementia, with metabolic diseases such as diabetes, hypertension and metabolic syndrome, as well as with some types of cancer (30). Exercise has both an anti-inflammatory effect through various pathways and reduces the secretion of inflammatory factors by reducing body fat mass and enhancing immune system function (31).
The exact mechanisms by which physical activity reduces inflammation are not fully understood. One of the possible mechanisms for the effects of exercise training on improving the inflammatory state is the intracellular signaling pathways mediated by nitric oxide (NO). Nitric oxide is closely related to the inflammatory state and acts as an important mediator of inflammation. The enzyme nitric oxide synthase (eNOS) regulates the expression of proinflammatory molecules such as NF-κB and cyclooxygenase 2 (COX2) as well as proinflammatory cytokines. Nitric oxide derived from eNOS plays an important role in some of the fundamental events of inflammation such as cell adhesion, modulation of vascular permeability, and angiogenesis. Notably, after moderate-intensity exercise, nitric oxide levels increase, leading to improved muscle function and inhibition of inflammation (32).
This study, conducted on an elderly population, faced inherent limitations regarding sample size, restricting us to 10 participants per group. Other limitations include the lack of measurement of additional inflammatory markers (e.g., TNF-α, IL-6) and the inability to control for potentially confounding environmental factors such as diet, sleep, and stress, which can influence inflammation.
Conclusion
This study demonstrated that an eight-week aerobic exercise program can reduce levels of the inflammatory marker cPLA2, potentially helping to prevent the onset of inflammation-related disorders in aging. Therefore, aerobic exercise can be considered a non-pharmacological strategy for mitigating aging-associated oxidative stress and enhancing the quality of life in the elderly.
Acknowledgements
This article is derived from the master's thesis of the Islamic Azad University of Ahvaz Branch, which hereby acknowledges and thanks the research deputy of the Islamic Azad University of Ahvaz Branch and the research participants.
Conflict of interest
The authors of the article declare that there is no conflict of interest in the present study.
Funding
This research received no external funding.
Authors' Contributions
Conceptualization: S.M., M.B.; Methodology: S.M., M.B.; Formal analysis: S.M., M.B.; Investigation: S.M., M.B.; Writing - original draft: S.M., M.B.; Writing–review & editing: S.M., M.B.; Supervision: M.B. All authors approved the final manuscript.
References
1. Singh A, Schurman SH, Bektas A, Kaileh M, Roy R, Wilson DM, et al. Aging and inflammation. Cold Spring Harbor Perspectives in Medicine. 2024; 14(6): 1-26.
2. Sun GY, Geng X, Teng T, Yang B, Appenteng MK, Greenlief CM, et al. Dynamic role of phospholipases A2 in health and diseases in the central nervous system. Cells. 2021; 10(11): 1-23.
3. Khan SA, Ilies MA. The phospholipase A2 superfamily: structure, isozymes, catalysis, physiologic and pathologic roles. International Journal of Molecular Sciences. 2023; 24(2): 1-35.
4. Hugo C, Asante I, Sadybekov A, Katritch V, Yassine HN. Development of calcium-dependent phospholipase a2 inhibitors to target cellular senescence and oxidative stress in neurodegenerative diseases. Antioxidants & Redox Signaling. 2024; 41(16-18): 1100-16.
5. Mukherjee AK. Correlation between the phospholipids domains of the target cell membrane and the extent of Naja kaouthia PLA2-induced membrane damage: evidence of distinct catalytic and cytotoxic sites in PLA2 molecules. Biochimica et Biophysica Acta (BBA)-General Subjects. 2007; 1770(2): 187–95.
6. Czapski GA, Czubowicz K, Strosznajder JB, Strosznajder RP. The lipoxygenases: their regulation and implication in Alzheimer’s disease. Neurochemical Research. 2016; 41(1): 243–57.
7. Strokin M, Sergeeva M, Reiser G. Docosahexaenoic acid and arachidonic acid release in rat brain astrocytes is mediated by two separate isoforms of phospholipase A2 and is differently regulated by cyclic AMP and Ca2+. British Journal of Pharmacology. 2003; 139(5): 1014-22.
8. Chao CC, Gutiérrez-Vázquez C, Rothhammer V, Mayo L, Wheeler MA, Tjon EC, et al. Metabolic control of astrocyte pathogenic activity via cPLA2-MAVS. Cell. 2019; 179(7): 1483–98.
9. Chuang DY, Simonyi A, Kotzbauer PT, Gu Z, Sun GY. Cytosolic phospholipase A2 plays a crucial role in ROS/NO signaling during microglial activation through the lipoxygenase pathway. Journal of Neuroinflammation. 2015; 12(1): 1-20.
10. Lee JCM, Simonyi A, Sun AY, Sun GY. Phospholipases A2 and neural membrane dynamics: implications for Alzheimer’s disease. Journal of Neurochemistry. 2011; 116(5): 813–9.
11. Gentile MT, Reccia M, Sorrentino P, Vitale E, Sorrentino G, Puca AA, et al. Role of cytosolic calcium-dependent phospholipase A2 in Alzheimer's disease pathogenesis. Molecular Neurobiology. 2012; 45(3): 596–604.
12. Gynther M, Estrada ML, Loppi S, Korhonen P, Kanninen KM, Malm T, et al. Increased expression and activity of brain cortical cPLA2 due to chronic lipopolysaccharide administration in mouse model of familial alzheimer’s disease. Pharmaceutics. 2022; 14(11): 1-12.
13. Ma QL, Ebright B, Li B, Li J, Galvan J, Sanchez A, et al. Evidence for cPLA2 activation in Alzheimer’s disease synaptic pathology. BioRxiv. 2025; 1: 1-43.
14. Kregel KC, Zhang HJ. An integrated view of oxidative stress in aging: basic mechanisms, functional effects, and pathological considerations. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology. 2007; 292(1): 18– 36.
15. Sun GY, Chuang DY, Zong Y, Jiang J, Lee JC, Gu Z, et al. Role of cytosolic phospholipase A2 in oxidative and inflammatory signaling pathways in different cell types in the central nervous system. Molecular Neurobiology. 2014; 50(1): 6–14.
16. Sanchez-Mejia RO, Newman JW, Toh S, Yu GQ, Zhou Y, Halabisky B, et al. Phospholipase A2 reduction ameliorates cognitive deficits in a mouse model of Alzheimer's disease. Nature Neuroscience. 2008; 11(11): 1311–8.
17. Bostani M, Noaein SA. The effect of continuous aerobic training on bax/bcl2 ratio in pancreatic tissue diabetic rats. Iranian Journal of Diabetes and Obesity. 2021.
18. Cerqueira É, Marinho DA, Neiva HP, Lourenço O. Inflammatory effects of high and moderate intensity exercise—a systematic review. Frontiers in Physiology. 2020; 10: 1-14.
19. de Sousa CV, Sales MM, Rosa TS, Lewis JE, de Andrade RV, Simoes HG. The antioxidant effect of exercise: a systematic review and meta-analysis. Sports Medicine. 2017; 47(2): 277–93.
20. de Souza DC, Matos VA, Dos Santos VO, Medeiros IF, Marinho CS, Nascimento PR, et al. Effects of high-intensity interval and moderate-intensity continuous exercise on inflammatory, leptin, IgA, and lipid peroxidation responses in obese males. Frontiers in Physiology. 2018; 9: 1-9.
21. Pillon Barcelos R, Freire Royes LF, Gonzalez-Gallego J, Bresciani G. Oxidative stress and inflammation: liver responses and adaptations to acute and regular exercise. Free Radical Research. 2017; 51(2): 222–36.
22. Bushman B, Medicine ACoS. ACSM's Complete Guide to Fitness & Health, 2E: Human Kinetics .2017.
23. Gómez-Rubio P, Trapero I. The beneficial effect of physical exercise on inflammatory makers in older individuals. Endocrine, Metabolic & Immune Disorders-Drug Targets . 2021; 21(6): 1008-16.
24. Buyukyazi G, Ulman C, Taneli F, Esen H, Gozlukaya F, Ozcan I, et al. The effects of different intensity walking programs on serum blood lipids, high-sensitive C-reactive protein, and lipoprotein-associated phospholipase A2 in premenopausal women. Science & Sports. 2010; 25(5): 245–52.
25. Thijssen DH, Cable NT, Green DJ. Impact of exercise training on arterial wall thickness in humans. Clinical Science. 2012; 122(7): 311–22.
26. Lavi S, McConnell JP, Rihal CS, Prasad A, Mathew V, Lerman LO, et al. Local production of lipoprotein-associated phospholipase A2 and lysophosphatidylcholine in the coronary circulation: association with early coronary atherosclerosis and endothelial dysfunction in humans. Circulation. 2007; 115(21): 2715–21.
27. Vina J, Borras C, Abdelaziz KM, Garcia-Valles R, Gomez-Cabrera MC. The free radical theory of aging revisited: the cell signaling disruption theory of aging. Antioxidants & Redox Signaling. 2013; 19(8): 779–87.
28. Kruk J, Aboul-Enein HY, Kładna A, Bowser JE. Oxidative stress in biological systems and its relation with pathophysiological functions: the effect of physical activity on cellular redox homeostasis. Free Radical Research. 2019; 53(5): 497–521.
29. Webb R, Hughes MG, Thomas AW, Morris K. The ability of exercise-associated oxidative stress to trigger redox-sensitive signalling responses. Antioxidants. 2017; 6(3): 1-21.
30. Deleidi M, Jäggle M, Rubino G. Immune aging, dysmetabolism, and inflammation in neurological diseases. Frontiers in Neuroscience. 2015;9: 1-14.
31. Wang S, Zhou H, Zhao C, He H. Effect of exercise training on body composition and inflammatory cytokine levels in overweight and obese individuals: a systematic review and network meta-analysis. Frontiers in Immunology. 2022; 13: 1-13.
32. Scheele C, Nielsen S, Pedersen BK. ROS and myokines promote muscle adaptation to exercise. Trends in Endocrinology & Metabolism. 2009; 20(3): 95–9.
Type of Study: Research |
Subject:
General
Received: 2025/10/8 | Accepted: 2025/11/30 | Published: 2025/12/19
Received: 2025/10/8 | Accepted: 2025/11/30 | Published: 2025/12/19
References
1. Singh A, Schurman SH, Bektas A, Kaileh M, Roy R, Wilson DM, et al. Aging and inflammation. Cold Spring Harbor Perspectives in Medicine. 2024; 14(6): 1-26.
2. Sun GY, Geng X, Teng T, Yang B, Appenteng MK, Greenlief CM, et al. Dynamic role of phospholipases A2 in health and diseases in the central nervous system. Cells. 2021; 10(11): 1-23.
3. Khan SA, Ilies MA. The phospholipase A2 superfamily: structure, isozymes, catalysis, physiologic and pathologic roles. International Journal of Molecular Sciences. 2023; 24(2): 1-35.
4. Hugo C, Asante I, Sadybekov A, Katritch V, Yassine HN. Development of calcium-dependent phospholipase a2 inhibitors to target cellular senescence and oxidative stress in neurodegenerative diseases. Antioxidants & Redox Signaling. 2024; 41(16-18): 1100-16.
5. Mukherjee AK. Correlation between the phospholipids domains of the target cell membrane and the extent of Naja kaouthia PLA2-induced membrane damage: evidence of distinct catalytic and cytotoxic sites in PLA2 molecules. Biochimica et Biophysica Acta (BBA)-General Subjects. 2007; 1770(2): 187–95.
6. Czapski GA, Czubowicz K, Strosznajder JB, Strosznajder RP. The lipoxygenases: their regulation and implication in Alzheimer’s disease. Neurochemical Research. 2016; 41(1): 243–57.
7. Strokin M, Sergeeva M, Reiser G. Docosahexaenoic acid and arachidonic acid release in rat brain astrocytes is mediated by two separate isoforms of phospholipase A2 and is differently regulated by cyclic AMP and Ca2+. British Journal of Pharmacology. 2003; 139(5): 1014-22.
8. Chao CC, Gutiérrez-Vázquez C, Rothhammer V, Mayo L, Wheeler MA, Tjon EC, et al. Metabolic control of astrocyte pathogenic activity via cPLA2-MAVS. Cell. 2019; 179(7): 1483–98.
9. Chuang DY, Simonyi A, Kotzbauer PT, Gu Z, Sun GY. Cytosolic phospholipase A2 plays a crucial role in ROS/NO signaling during microglial activation through the lipoxygenase pathway. Journal of Neuroinflammation. 2015; 12(1): 1-20.
10. Lee JCM, Simonyi A, Sun AY, Sun GY. Phospholipases A2 and neural membrane dynamics: implications for Alzheimer’s disease. Journal of Neurochemistry. 2011; 116(5): 813–9.
11. Gentile MT, Reccia M, Sorrentino P, Vitale E, Sorrentino G, Puca AA, et al. Role of cytosolic calcium-dependent phospholipase A2 in Alzheimer's disease pathogenesis. Molecular Neurobiology. 2012; 45(3): 596–604.
12. Gynther M, Estrada ML, Loppi S, Korhonen P, Kanninen KM, Malm T, et al. Increased expression and activity of brain cortical cPLA2 due to chronic lipopolysaccharide administration in mouse model of familial alzheimer’s disease. Pharmaceutics. 2022; 14(11): 1-12.
13. Ma QL, Ebright B, Li B, Li J, Galvan J, Sanchez A, et al. Evidence for cPLA2 activation in Alzheimer’s disease synaptic pathology. BioRxiv. 2025; 1: 1-43.
14. Kregel KC, Zhang HJ. An integrated view of oxidative stress in aging: basic mechanisms, functional effects, and pathological considerations. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology. 2007; 292(1): 18– 36.
15. Sun GY, Chuang DY, Zong Y, Jiang J, Lee JC, Gu Z, et al. Role of cytosolic phospholipase A2 in oxidative and inflammatory signaling pathways in different cell types in the central nervous system. Molecular Neurobiology. 2014; 50(1): 6–14.
16. Sanchez-Mejia RO, Newman JW, Toh S, Yu GQ, Zhou Y, Halabisky B, et al. Phospholipase A2 reduction ameliorates cognitive deficits in a mouse model of Alzheimer's disease. Nature Neuroscience. 2008; 11(11): 1311–8.
17. Bostani M, Noaein SA. The effect of continuous aerobic training on bax/bcl2 ratio in pancreatic tissue diabetic rats. Iranian Journal of Diabetes and Obesity. 2021.
18. Cerqueira É, Marinho DA, Neiva HP, Lourenço O. Inflammatory effects of high and moderate intensity exercise—a systematic review. Frontiers in Physiology. 2020; 10: 1-14.
19. de Sousa CV, Sales MM, Rosa TS, Lewis JE, de Andrade RV, Simoes HG. The antioxidant effect of exercise: a systematic review and meta-analysis. Sports Medicine. 2017; 47(2): 277–93.
20. de Souza DC, Matos VA, Dos Santos VO, Medeiros IF, Marinho CS, Nascimento PR, et al. Effects of high-intensity interval and moderate-intensity continuous exercise on inflammatory, leptin, IgA, and lipid peroxidation responses in obese males. Frontiers in Physiology. 2018; 9: 1-9.
21. Pillon Barcelos R, Freire Royes LF, Gonzalez-Gallego J, Bresciani G. Oxidative stress and inflammation: liver responses and adaptations to acute and regular exercise. Free Radical Research. 2017; 51(2): 222–36.
22. Bushman B, Medicine ACoS. ACSM's Complete Guide to Fitness & Health, 2E: Human Kinetics .2017.
23. Gómez-Rubio P, Trapero I. The beneficial effect of physical exercise on inflammatory makers in older individuals. Endocrine, Metabolic & Immune Disorders-Drug Targets . 2021; 21(6): 1008-16.
24. Buyukyazi G, Ulman C, Taneli F, Esen H, Gozlukaya F, Ozcan I, et al. The effects of different intensity walking programs on serum blood lipids, high-sensitive C-reactive protein, and lipoprotein-associated phospholipase A2 in premenopausal women. Science & Sports. 2010; 25(5): 245–52.
25. Thijssen DH, Cable NT, Green DJ. Impact of exercise training on arterial wall thickness in humans. Clinical Science. 2012; 122(7): 311–22.
26. Lavi S, McConnell JP, Rihal CS, Prasad A, Mathew V, Lerman LO, et al. Local production of lipoprotein-associated phospholipase A2 and lysophosphatidylcholine in the coronary circulation: association with early coronary atherosclerosis and endothelial dysfunction in humans. Circulation. 2007; 115(21): 2715–21.
27. Vina J, Borras C, Abdelaziz KM, Garcia-Valles R, Gomez-Cabrera MC. The free radical theory of aging revisited: the cell signaling disruption theory of aging. Antioxidants & Redox Signaling. 2013; 19(8): 779–87.
28. Kruk J, Aboul-Enein HY, Kładna A, Bowser JE. Oxidative stress in biological systems and its relation with pathophysiological functions: the effect of physical activity on cellular redox homeostasis. Free Radical Research. 2019; 53(5): 497–521.
29. Webb R, Hughes MG, Thomas AW, Morris K. The ability of exercise-associated oxidative stress to trigger redox-sensitive signalling responses. Antioxidants. 2017; 6(3): 1-21.
30. Deleidi M, Jäggle M, Rubino G. Immune aging, dysmetabolism, and inflammation in neurological diseases. Frontiers in Neuroscience. 2015;9: 1-14.
31. Wang S, Zhou H, Zhao C, He H. Effect of exercise training on body composition and inflammatory cytokine levels in overweight and obese individuals: a systematic review and network meta-analysis. Frontiers in Immunology. 2022; 13: 1-13.
32. Scheele C, Nielsen S, Pedersen BK. ROS and myokines promote muscle adaptation to exercise. Trends in Endocrinology & Metabolism. 2009; 20(3): 95–9.
Send email to the article author
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |







